PIANO
PIANO (Pregnancy Inflammatory bowel disease And Neonatal Outcomes) is a national study of women with IBD and their children. This study looks at the safety of IBD medications on the pregnancy and short- and long-term outcomes of the children.
Purpose
Determine the safety of IBD medications on pregnancy outcomes and impact on children exposed in utero.
What are we measuring:
- Whether the level of biologic drug transferred across the placenta to the infant by the time of birth predicts the risk of infection or other adverse outcomes
- Whether the achievement of developmental milestones is affected by medication exposure
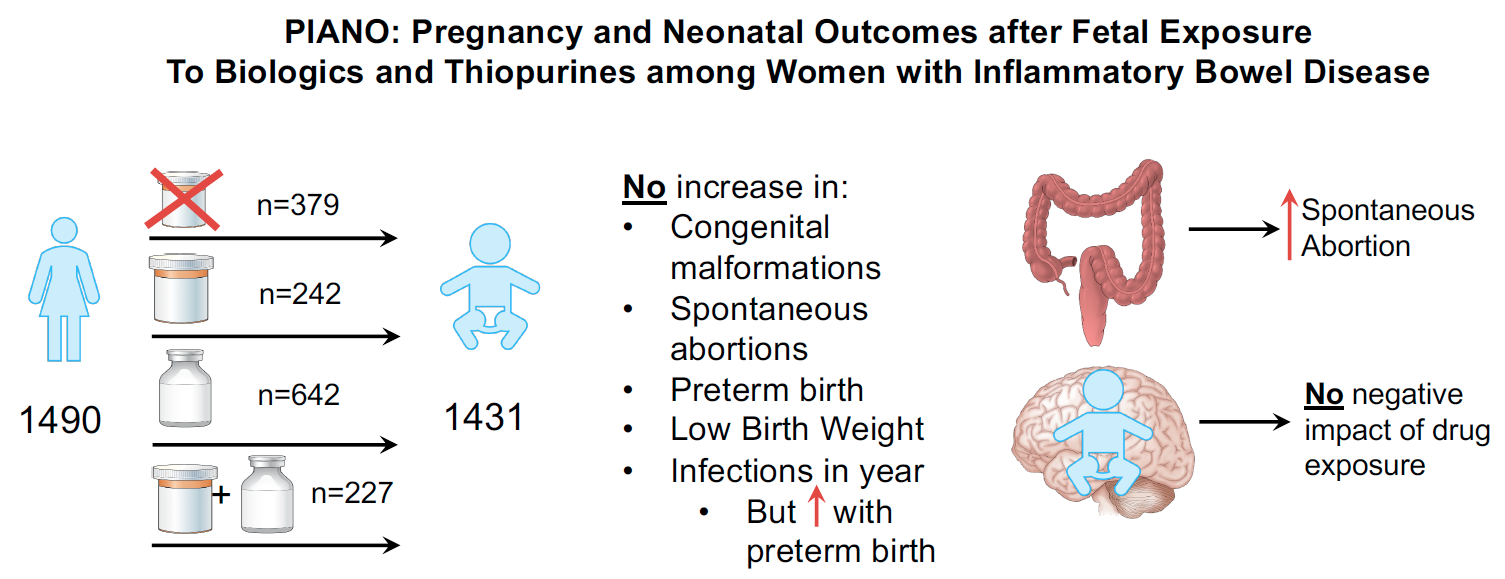
- Whether the rates of birth defects, adverse pregnancy outcomes and complications of labor and delivery are affected by IBD medications
- Whether second trimester drug levels can be used to adjust drug and minimize transfer across the placenta to the baby
- What are the outcomes of children out to 18 years of age based on exposure to IBD Medications in the womb
- Do Covid vaccine generated antibodies cross to the baby from the mother as measured by levels in the cord blood at birth
Little is known about the effects of azathioprine/6MP or biologic therapy (Remicade, Humira, Cimzia, Tysabri, Stelara, Simponi, Entyvio) and small molecules (Xeljanz, Zeposia, Rinvoq) on pregnant women and their babies. As many women receive these during their prime reproductive years, the information from PIANO will be valuable in guiding therapy of women with CD or UC who wish to have children while receiving this therapy for their illness.
Please see below our current results and outcomes. If you are interested in enrolling please call 415-885-3734 or email [email protected]
What the Study Entails
The PIANO study mainly gathers information through questionnaires. We ask that you complete a questionnaire at each trimester of your pregnancy, at the end of your pregnancy, 4, 9, and 12 months post-delivery, and once a year up to the age of 18 years of age. You may stop at any time.
We also ask for blood work at your 2nd trimester as well as blood work from you and your umbilical cord at delivery.
All materials that are needed will be sent to you via FedEx so you will not have to worry about finding material or having to provide it on your own.
Donate
If you would like to donate to the PIANO study, please click the "MAKE A GIFT" button below. Any amount of contribution will be greatly appreciated and will help further our understanding of pregnancy and IBD.