Clinical Trials
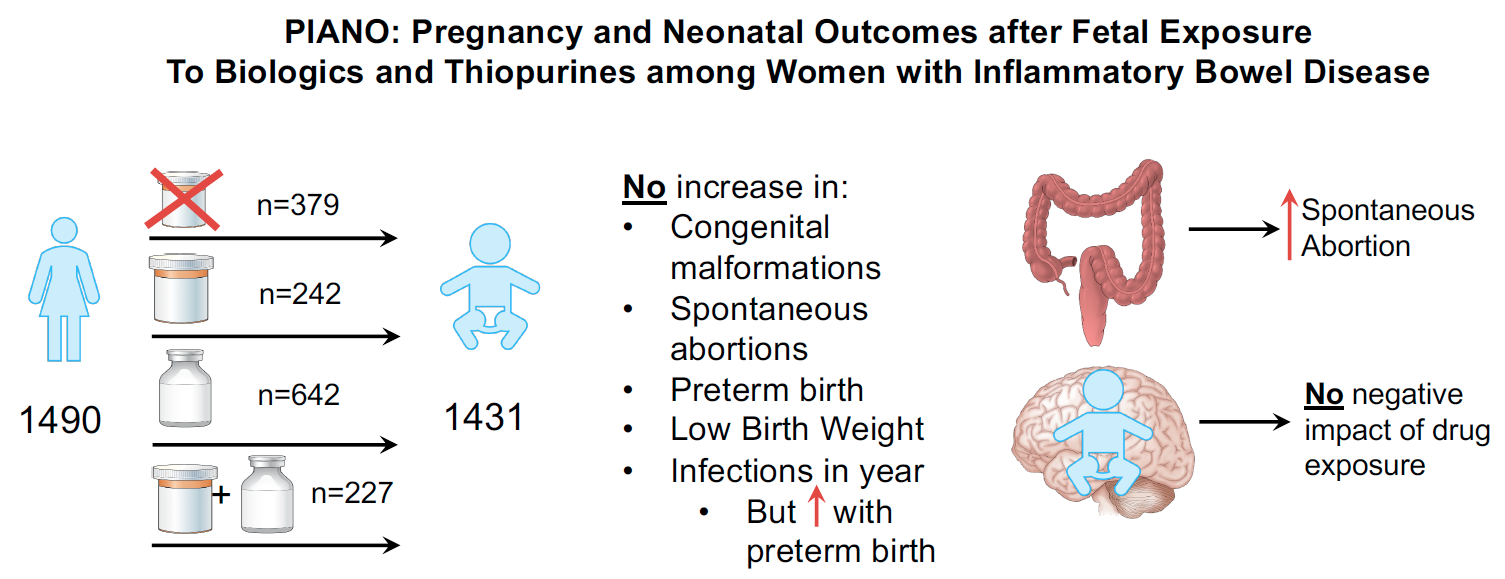
PIANO: A national pregnancy registry of women with IBD
This national prospective registry study is conducted to determine whether the rates of birth defects, miscarriages, premature births and other outcomes in women with inflammatory bowel disease (IBD) taking azathioprine/6MP or biologic therapy (Remicade, Humira, Cimzia, Tysabri, Stelara, Simponi, Entyvio, or Xeljanz) are different from those among IBD-affected women not taking these medications.
For more information, please visit the PIANO website.
![]() PIANO Registry (pdf)
PIANO Registry (pdf)
SIBERITE: RO7790121 in Patients With Moderately to Severely Active Crohn's Disease
The SIBERITE study is a Phase III clinical trial by Genentech evaluating RO7790121, a potential treatment for moderately to severely active Crohn's Disease (CD). This drug targets TL1A, a protein involved in gut inflammation, to help reduce symptoms and maintain remission. The study is enrolling adults (16-80) who have not responded well to other therapies. By participating, individuals contribute to research that may improve future CD therapies.
For more information, please view the ICF below:
PALEKONA: GS-5290 in Participants With Moderately to Severely Active Ulcerative Colitis
The PALEKONA study is a Phase 2 clinical trial by Gilead evaluating tilpisertib fosmecarbil (GS-5290), an investigational drug designed to block Syk, a protein involved in inflammation, in adults with moderately to severely active ulcerative colitis. Participants will take either the study drug or a placebo for 12 weeks, with the option to continue treatment for up to 64 weeks.
For more information, please view the ICF below:
REASON: Study to Evaluate Transmural Healing and Disease
Modifying Effect of Guselkumab in Crohn’s Disease Patients
The REASON study is a Phase 3b, open-label, multicenter clinical trial sponsored by Janssen evaluating TREMFYA (guselkumab) in adults with Crohn’s disease. This study investigates how guselkumab, which targets the IL-23 pathway, may promote transmural healing and modify disease progression. To assess treatment response, the study uses advanced imaging tools including intestinal ultrasound (IUS) and magnetic resonance enterography (MRE). By participating, individuals help advance research into potential new treatment options for Crohn’s disease.
For more information, please view the ICF below:
